Worm Lab
Day 1:
On the first day when we were introduced to this project, we created a T chart that organized our data into Observation/Questions/Research. We looking into the microscopes and saw hundreds of worms, after watching them for a while, there was no movement. But then after looking into another scope that had better magnifying, I could see the worms very close up and I could see a lot more. I then did research on the worms and what they are used for.
Day 2:
On the second day we were told to go outside and collect dirt from two different locations. I saw people grabbing dry dirt and even sand. I thought about if I was a worm where would I wanna live. I thought about a moist shady place with a food source near by. So I looked around for a shady place, once I found a spot I looked around to find any trash on the ground. I saw a small orange peel laying in the dirt and I thought that there would be worms around it so I collected the orange peel and collected the dirt surrounding it. The next place I look was under a rock because worms like to live where its nice and cool. So I flipped over a huge rock and collected dirt underneath it.
Day 3:
After reviewing my dirt under a microscope I was sad to see there was no nematodes....
On the first day when we were introduced to this project, we created a T chart that organized our data into Observation/Questions/Research. We looking into the microscopes and saw hundreds of worms, after watching them for a while, there was no movement. But then after looking into another scope that had better magnifying, I could see the worms very close up and I could see a lot more. I then did research on the worms and what they are used for.
Day 2:
On the second day we were told to go outside and collect dirt from two different locations. I saw people grabbing dry dirt and even sand. I thought about if I was a worm where would I wanna live. I thought about a moist shady place with a food source near by. So I looked around for a shady place, once I found a spot I looked around to find any trash on the ground. I saw a small orange peel laying in the dirt and I thought that there would be worms around it so I collected the orange peel and collected the dirt surrounding it. The next place I look was under a rock because worms like to live where its nice and cool. So I flipped over a huge rock and collected dirt underneath it.
Day 3:
After reviewing my dirt under a microscope I was sad to see there was no nematodes....
The watershed Lab Write up
Eli Steria
Jennifer Buffington, Gillian Hooper
December 14, 2014
The goal of this experiment was to see how the shrimp react in different amounts of light and darkness.
We put black clothes over the tanks to create darkness for the shrimp to see if they like it. Also we put nothing over the tank and just had the lights in the room reflect in the tank and see how they liked it.
Data Tables
With Light:
12-9-14
-Shrimp are staying along the bottom.
-Seem to move around and not stay in one place.
12-10-14
-Climbing on rocks hanging out their not doing much.
12-11-14
-Moving around, looking through rocks.
Changed to Darkness:
12-12-14
- Staying around the ground not moving much (but they aren’t dead).
12-15-14
-Staying in one place running away from light.
12-16-14
-Staying in one place, not moving much.
I think we did not have enough time for this lab. As a group we would have liked to put them through extreme light, but never got a chance to. Next time I would either shorten the days we studied in each amount of light to be able to squeeze all three in the time allowed.
Towards the end I found that our hypothesis was right that they would move around in the light, but I wish we would have gotten a chance to try extreme light, but they enjoyed moving around and digging through the rocks throughout the normal light days, but seemed to not move much when they were put in the dark. This lab was something I have never done and I learned a little bit about animal behavior.
Jennifer Buffington, Gillian Hooper
December 14, 2014
- Would you like light or no light Mr. Shrimp?
The goal of this experiment was to see how the shrimp react in different amounts of light and darkness.
We put black clothes over the tanks to create darkness for the shrimp to see if they like it. Also we put nothing over the tank and just had the lights in the room reflect in the tank and see how they liked it.
Data Tables
With Light:
12-9-14
-Shrimp are staying along the bottom.
-Seem to move around and not stay in one place.
12-10-14
-Climbing on rocks hanging out their not doing much.
12-11-14
-Moving around, looking through rocks.
Changed to Darkness:
12-12-14
- Staying around the ground not moving much (but they aren’t dead).
12-15-14
-Staying in one place running away from light.
12-16-14
-Staying in one place, not moving much.
I think we did not have enough time for this lab. As a group we would have liked to put them through extreme light, but never got a chance to. Next time I would either shorten the days we studied in each amount of light to be able to squeeze all three in the time allowed.
Towards the end I found that our hypothesis was right that they would move around in the light, but I wish we would have gotten a chance to try extreme light, but they enjoyed moving around and digging through the rocks throughout the normal light days, but seemed to not move much when they were put in the dark. This lab was something I have never done and I learned a little bit about animal behavior.
Flame Test lab
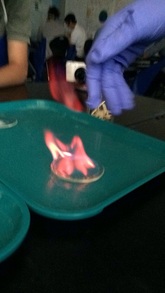
Eli Steria
Joceyln Villa
09-10-2014
Purpose: This lab was an experience with chemicals and seeing what color it would be if you mixed it with methanol and lit it.
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
Electrons around the nucleus of the metal atoms in these chemicals must be moved to a higher energy state from the heat in the flame.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
Nonmetals can form different types of bonds depending on their partner atoms.
3. What is the definition of a salt in chemistry?
A salt is the name that is given to a product of a reaction between an acid and a base.
4. What is fire?
Fire is the rapid mixed of oxygen with fuel when there is heat, a body of incandescent gas that contains and sustains the reaction and emits light and heat.
5. What are the chemicals involved in lighting a match?
The top of the match is pretty much sulfur and potassium chlorate with powdered glass, colorants, fillers, and a binder made of glue and starch. When striking the box consists of powdered glass o, red phosphorus, binder, and filler. When you strike a safety match, the glass-on-glass friction generates heat, converting a small amount of red phosphorus to white phosphorus vapor.
6. What is a chemical reaction?
7. What natural cycles does wildfire have an impact on?
Joceyln Villa
09-10-2014
Purpose: This lab was an experience with chemicals and seeing what color it would be if you mixed it with methanol and lit it.
1. Why do you think the chemicals have to be heated in the flame first before the colored light is emitted?
Electrons around the nucleus of the metal atoms in these chemicals must be moved to a higher energy state from the heat in the flame.
2. Most salts contain a metal and a non-metal. What type of bonds usually form between a metal and a non-metal?
Nonmetals can form different types of bonds depending on their partner atoms.
3. What is the definition of a salt in chemistry?
A salt is the name that is given to a product of a reaction between an acid and a base.
4. What is fire?
Fire is the rapid mixed of oxygen with fuel when there is heat, a body of incandescent gas that contains and sustains the reaction and emits light and heat.
5. What are the chemicals involved in lighting a match?
The top of the match is pretty much sulfur and potassium chlorate with powdered glass, colorants, fillers, and a binder made of glue and starch. When striking the box consists of powdered glass o, red phosphorus, binder, and filler. When you strike a safety match, the glass-on-glass friction generates heat, converting a small amount of red phosphorus to white phosphorus vapor.
6. What is a chemical reaction?
7. What natural cycles does wildfire have an impact on?
The water cycle, carbon cycle and nitrogen cycle.
8. List at least three impacts of wildfire in San Diego.
8. List at least three impacts of wildfire in San Diego.
Wildlife, people homes,
sAVE fred
Eli Steria
Zoe
8/25/2014
The purpose of the Save Fred project, was to get the life perserver on to Fred to save him from falling in to the water. My idea to save Fred is to have Zoe hold Fred with a Fred Toucher. (Paper Clip) Then I was use the paper clip to flip the cup upside down so then the life perserver (Peach Ring) is now open to pick up. Then I would use the same paperclip I used to flip the cup to pick up the life preserver. Then me and my partner Zoe would have to work together and slide the life preserver on to Fred.
Pre-Lab Questions:
1. Why is it important in science to follow an agreed-upon procedure?
If someone in the group was not on the same page as the others, something could seriously go wrong. Maybe he/she isn't aware on what their part is in the project and they could completely not do a part of the project and everyones grade has to suffer from he/she's mistake.
2. Why is important to document you procedure and any errors or differences?
You can keep of information for future observations on other projects.
3. Why do we not normally have food in Chemistry Class?
It attracts ants, plus you never know what chemical was there last. Food and Chemistry don't mix.
Measurements:
Gummy Worm:
Length: 8 cm.
Mass: 7.9 g
Live Vest:
Mass: 7,700 mg
Diameter: 35mm
Boat:
Volume: 266 mm
Circumference: 2664 cm
Zoe
8/25/2014
The purpose of the Save Fred project, was to get the life perserver on to Fred to save him from falling in to the water. My idea to save Fred is to have Zoe hold Fred with a Fred Toucher. (Paper Clip) Then I was use the paper clip to flip the cup upside down so then the life perserver (Peach Ring) is now open to pick up. Then I would use the same paperclip I used to flip the cup to pick up the life preserver. Then me and my partner Zoe would have to work together and slide the life preserver on to Fred.
Pre-Lab Questions:
1. Why is it important in science to follow an agreed-upon procedure?
If someone in the group was not on the same page as the others, something could seriously go wrong. Maybe he/she isn't aware on what their part is in the project and they could completely not do a part of the project and everyones grade has to suffer from he/she's mistake.
2. Why is important to document you procedure and any errors or differences?
You can keep of information for future observations on other projects.
3. Why do we not normally have food in Chemistry Class?
It attracts ants, plus you never know what chemical was there last. Food and Chemistry don't mix.
Measurements:
Gummy Worm:
Length: 8 cm.
Mass: 7.9 g
Live Vest:
Mass: 7,700 mg
Diameter: 35mm
Boat:
Volume: 266 mm
Circumference: 2664 cm
MEASUREMENTS
Erlenmeyer: 260 ML
Bottle: 380 ML
Conical: 100 ML
400 ML Beaker: 33 ML
10 ML Cylinder: 6ML
250 ML Cylinder: 15 ML
10 ML Beaker: 8 ML
Volumetric Flask: 150 ML
Flask: 1260 ML
600 ML Beaker: 20 F
400 ML Beaker: 70 F
Length of Tray: 0.4 M
Length of Blue Dots: 4.5 cm
Mass of 2 Shells: 1.4 G
Mass of 1 Shell: .9 G
This Lab was easy but it got a little confusing at some parts making sure I was measuring/weighing in the right unit. At some points I would check if my answers were right by asking other students what they got and sometimes I would be incorrect. So I would have to go back and re-measure.
Below is sketches for each measurement in order:
Bottle: 380 ML
Conical: 100 ML
400 ML Beaker: 33 ML
10 ML Cylinder: 6ML
250 ML Cylinder: 15 ML
10 ML Beaker: 8 ML
Volumetric Flask: 150 ML
Flask: 1260 ML
600 ML Beaker: 20 F
400 ML Beaker: 70 F
Length of Tray: 0.4 M
Length of Blue Dots: 4.5 cm
Mass of 2 Shells: 1.4 G
Mass of 1 Shell: .9 G
This Lab was easy but it got a little confusing at some parts making sure I was measuring/weighing in the right unit. At some points I would check if my answers were right by asking other students what they got and sometimes I would be incorrect. So I would have to go back and re-measure.
Below is sketches for each measurement in order:

