10th Grade final exibition
|
If we had more time and money it would of been nice to do my project pitch instead. To create a small version of my idea would of been great. We could of created a pump system and everything but my pitch was very expensive to actually build. I had a great time building Christians project and enjoyed testing it. I would defiantly do this project again!
Site: trawabo.weebly.com Inscrutable: http://www.instructables.com/id/Charclish/ |
Students without Borders project pitch
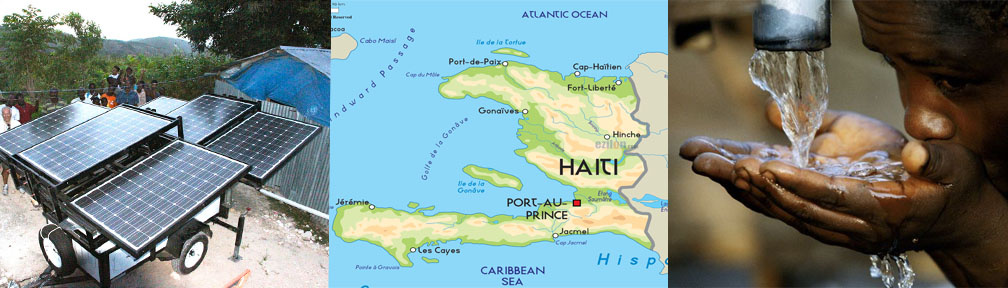
Haiti is a place that I am highly interested in after that huge earthquake with a magnitude of 7.0 in 2010. Everyday accommodations that we take for granted they don’t have. Such as clean environments, bathrooms, fresh clean water, shelter, etc. I want to go into the infrastructure of water supply in Haiti. The shallow hand dug wells that exit there currently are polluted because they are open and contaminated water from local rivers spills back into them. Ground water wells must be protected so they cannot become contaminated. Since electricity isn't available over there, we would have to use different source of power. Thats why I chose using solar power that uses DC power. The solar panels would turn on for two hours of the day producing about 2,400 gallons of water. The cost of 1050 watts of Solar Panels is $5130, cost of Pumps about the same plus we need to dig a well and cover it with concrete so it can help protect the water from being contaminated. There would also be a reservoir to storage the water so if there isn't any sun for the solar panel they will still have water. There would also be a supplementary power source. They will contain sufficient energy to power the system for a maximum of two days without a recharge. The total cost would be in the $20,000 range. This is highly expensive but donations and fundraising I think it is very possible to achieve this amount of money.
The Watershed Project Reflection
The greatest strength of my exhibition project was the video I created with Frank, Lee Lee and Zoey of the San Marcos Watershed. It was creative and had a lot of great information that was interesting to the viewers. I knew it was a good video since a lot of people were attracted to the screen and watched the video throughly and gave me great feedback.
The thing I would most like to have improved about my exhibition project is the shrimp lab. I think we didn't spend enough time on our experiments to be personally honest, I spent a little amount of time actually looking at my shrimp. My other group members were in charge of taking notes and evaluating them depending on how much light they had in there tank. I think if we had more time I would of helped more in that part of the project.
Before the exhibition project even started I really didn't know what a watershed was. The whole project was a big learning curve for me personally. I learned a lot of new information including what is a watershed, where it is, and how we can help it. I think the most important thing I learned was when I did an interview with the Indiana Farmer with a huge chicken farm. I was able to touch one of his nerves and get information out of him that was very interesting. I learned how to be a good interviewee and how to connect with a human through an interview.
The thing I would most like to have improved about my exhibition project is the shrimp lab. I think we didn't spend enough time on our experiments to be personally honest, I spent a little amount of time actually looking at my shrimp. My other group members were in charge of taking notes and evaluating them depending on how much light they had in there tank. I think if we had more time I would of helped more in that part of the project.
Before the exhibition project even started I really didn't know what a watershed was. The whole project was a big learning curve for me personally. I learned a lot of new information including what is a watershed, where it is, and how we can help it. I think the most important thing I learned was when I did an interview with the Indiana Farmer with a huge chicken farm. I was able to touch one of his nerves and get information out of him that was very interesting. I learned how to be a good interviewee and how to connect with a human through an interview.
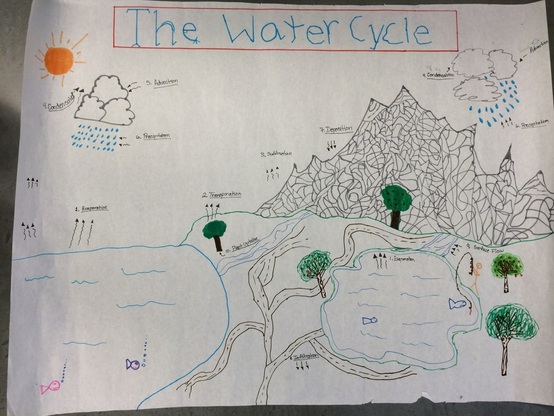
The WATER CYCLE

The water cycle is a constant cycle of the water on top, above and below the surface of the earth. It goes through 6 main cycles including evaporation, condensation, precipitation, collection, transpiration, and sublimation. Evaporation is the cause from the heat from the sun to evaporate the water in lakes, oceans, etc. to rise to the sky. The water vapor collects in the sky and forms clouds. Condensation causes the water vapor in the clouds to cool down and become water again. Precipitation is simply when rain, snow, hail, and sleet falls from the sky. Collection just collects water from the sky and forms a lake or ocean. Then it evaporates in the sky and then the cycle continues. Transpiration is like sweating, plants lose water which is absorbed into the atmosphere similar to evaporation. Sublimation is when solid to transform into a gas directly without becoming a liquid. Dry ice is the most common example of sublimation which sublimes at normal air temperature.
Water is the chemical substance that has the chemical formula H2O: one molecule of water has two hydrogen atom that share the electrons between atoms bonded to a single oxygen atom. Water’s chemical name is Dihydrogen Monoxide Hoax. Water is 3 atoms which is 2 hydrogen and 1 oxygen that is H2O.
The ocean holds 97% of the earths water. The remaining 3% of the water is freshwater which remains in glaciers, below the ground and ice.
Residence time is the time the particle spends in a particular place. Such in a river channel it last for 2 weeks, oceans can last up to 4,000 years and even glaciers can last anywhere from 1,000 to 10,000 years. The average time water spends in the atmosphere is 1 and half weeks. Biospheric is where the water stays inside plants and the average time spent in a plant is 1 week.
An example of the water cycle in San Diego could be any river, lake, pond, stream, creek, ocean, etc. Such at Lakes hodges, water evaporates into the sky and creates clouds. Then once it will become cool and turn the water vapor to water then it will rain. Then once again it will collect and fill an area and then the cycle will continue.
Sources:
http://www.sciencekids.co.nz/sciencefacts/weather/thewatercycle.html
http://www.unep.org/dewa/vitalwater/article26.html
Ionic & Covalent Bonding Research
Eli Steria
Ionic Bonding:
1. What types of atoms typically form ionic bonds?
An Ionic bond is formed by metals and nonmetals. Any metals in the reference table can form an ionic bond with an atom. For example, Lithium, Sodium and Calcium.
2. How are ionic bonds formed and what type of structure do they create?
When atoms get close together, sometimes they form ionic bonds that stick them together into molecules. These ionic bonds are weaker than covalent bonds, but they do still hold the molecules together.
3. What are the typical properties of ionic substances. Including the following: physical state, melting point, solubility in water, electrical conductivity, and any other properties you’d like to include.
1. Ionic compounds form crystals.
Molecular compounds form crystals, they frequently take other forms plus molecular crystals typically are softer than ionic crystals.
2. Ionic compounds have high melting points and high boiling points.
High temperatures are needed to separate positive and negative ions in ionic compounds. A lot of energy is required to melt ionic compounds.
3. Ionic compounds have higher enthalpies of fusion and vaporization than molecular compounds.
They have a lot higher melting and boiling points. It have enthalpies of fusion and vaporization that can be 10 to 100 times higher in temperature than those of most molecular compounds. The enthalpy of fusion is the heat needed to melt a single mole of a solid under constant pressure. The enthalpy of vaporization is the heat required for vaporize one mole of a liquid compound under constant pressure.
4. Ionic compounds are hard and brittle.
Ionic crystals are hard because the positive and negative ions are strongly attracted to each other and are very hard to take apart but when you add pressure to an ionic crystal the ions of charge can be forced closer to each other. ionic solids are very brittle.
5. Ionic compounds conduct electricity when they are dissolved in water.
When ionic compounds are dissolved in to water the ions separates and are free to conduct electric charge through the solution. Molten salts also create electricity.
6.Ionic solids are good insulators.
Although they conduct in molten form or in aqueous solution, ionic solids do not conduct electricity very well because the ions are bound so tightly to each other.
Image that represents ionic bonding:
Covalent Bonding:
What types of atoms typically form covalent bonds?
A pair on the oxygen atom in water contributes two electrons to make a covalent bond with a hydrogen ion to form the hydronium ion.
How are covalent bonds formed and what types of structure do they create?
Covalent bonds are formed by electron sharing between atoms. They create structures called molecules.
What are the typical properties of covalent substances? Include the following: physical state, melting point, solubility in water, electrical conductivity and any other properties you’d like to include.
Covalent compounds generally have much lower melting and boiling points that ionic compounds.
Covalent compounds are soft and squishy.
Covalent compounds tend to be more flammable that ionic compounds.
Covalent compounds don't conduct electricity in water.
Covalent compounds aren't usually very soluble in water.
Explain the difference between polar and nonpolar covalent bonds. Be sure to describe electronegative and the role it plays in determining which type of bond is formed.
Create an image that you feel best represents covalent bonding.
If two atoms have similar electronegativity, they will attract and share electrons equally. The average electrons spend the same amount of time next to each atom, so the electrons' negative charge is equally across the bond. This results in a non-polar covalent bond.
If one atom has an advantage over the other then the electrons are more attracted to that atom and spend more time at that end of the bond. That end of the bond ends up with a partial negative charge, thanks to the extra electron being there. The atom at the other end of the bond sees the electrons for a smaller amount of time, and will have a partial positive charge. With two oppositely charged ends, the bond is now designated polar covalent.
Story:
A Day in the Life
Once there was atom named Jimmy and another atom named Bob. There are in 6th grade, it was lunch time and Jimmy forget his lunch. Bob, a tiny blond little boy was sitting by himself at a blue table eating an electron sandwich. Jimmy came over and told him to give him his lunch, Bob said no way. After arguing for a while, it was the hottest point of the day. It reached a boiling point and both Jimmy and bob melted and turned into sharp crystals. They kept fighting, until the crystal broke because they were so brittle. They returned back to atoms and there grab onto one another and rolled down the hill into a fountain in the front of the school. Then they both dissolved, the atoms were stuck in the fountain until winter when the janitor drained out the fountain in the winter. Both Jimmy and Bob were stuck out in the snow but at least they were both good insulators until Jimmy reached over and grabbed Bob’s moldy old half eaten electron sandwich
A Day in the Life
Once there was a atom named Trip and Daniel. There were both in search of an electron house.
Ionic Bonding:
1. What types of atoms typically form ionic bonds?
An Ionic bond is formed by metals and nonmetals. Any metals in the reference table can form an ionic bond with an atom. For example, Lithium, Sodium and Calcium.
2. How are ionic bonds formed and what type of structure do they create?
When atoms get close together, sometimes they form ionic bonds that stick them together into molecules. These ionic bonds are weaker than covalent bonds, but they do still hold the molecules together.
3. What are the typical properties of ionic substances. Including the following: physical state, melting point, solubility in water, electrical conductivity, and any other properties you’d like to include.
1. Ionic compounds form crystals.
Molecular compounds form crystals, they frequently take other forms plus molecular crystals typically are softer than ionic crystals.
2. Ionic compounds have high melting points and high boiling points.
High temperatures are needed to separate positive and negative ions in ionic compounds. A lot of energy is required to melt ionic compounds.
3. Ionic compounds have higher enthalpies of fusion and vaporization than molecular compounds.
They have a lot higher melting and boiling points. It have enthalpies of fusion and vaporization that can be 10 to 100 times higher in temperature than those of most molecular compounds. The enthalpy of fusion is the heat needed to melt a single mole of a solid under constant pressure. The enthalpy of vaporization is the heat required for vaporize one mole of a liquid compound under constant pressure.
4. Ionic compounds are hard and brittle.
Ionic crystals are hard because the positive and negative ions are strongly attracted to each other and are very hard to take apart but when you add pressure to an ionic crystal the ions of charge can be forced closer to each other. ionic solids are very brittle.
5. Ionic compounds conduct electricity when they are dissolved in water.
When ionic compounds are dissolved in to water the ions separates and are free to conduct electric charge through the solution. Molten salts also create electricity.
6.Ionic solids are good insulators.
Although they conduct in molten form or in aqueous solution, ionic solids do not conduct electricity very well because the ions are bound so tightly to each other.
Image that represents ionic bonding:
Covalent Bonding:
What types of atoms typically form covalent bonds?
A pair on the oxygen atom in water contributes two electrons to make a covalent bond with a hydrogen ion to form the hydronium ion.
How are covalent bonds formed and what types of structure do they create?
Covalent bonds are formed by electron sharing between atoms. They create structures called molecules.
What are the typical properties of covalent substances? Include the following: physical state, melting point, solubility in water, electrical conductivity and any other properties you’d like to include.
Covalent compounds generally have much lower melting and boiling points that ionic compounds.
Covalent compounds are soft and squishy.
Covalent compounds tend to be more flammable that ionic compounds.
Covalent compounds don't conduct electricity in water.
Covalent compounds aren't usually very soluble in water.
Explain the difference between polar and nonpolar covalent bonds. Be sure to describe electronegative and the role it plays in determining which type of bond is formed.
Create an image that you feel best represents covalent bonding.
If two atoms have similar electronegativity, they will attract and share electrons equally. The average electrons spend the same amount of time next to each atom, so the electrons' negative charge is equally across the bond. This results in a non-polar covalent bond.
If one atom has an advantage over the other then the electrons are more attracted to that atom and spend more time at that end of the bond. That end of the bond ends up with a partial negative charge, thanks to the extra electron being there. The atom at the other end of the bond sees the electrons for a smaller amount of time, and will have a partial positive charge. With two oppositely charged ends, the bond is now designated polar covalent.
Story:
A Day in the Life
Once there was atom named Jimmy and another atom named Bob. There are in 6th grade, it was lunch time and Jimmy forget his lunch. Bob, a tiny blond little boy was sitting by himself at a blue table eating an electron sandwich. Jimmy came over and told him to give him his lunch, Bob said no way. After arguing for a while, it was the hottest point of the day. It reached a boiling point and both Jimmy and bob melted and turned into sharp crystals. They kept fighting, until the crystal broke because they were so brittle. They returned back to atoms and there grab onto one another and rolled down the hill into a fountain in the front of the school. Then they both dissolved, the atoms were stuck in the fountain until winter when the janitor drained out the fountain in the winter. Both Jimmy and Bob were stuck out in the snow but at least they were both good insulators until Jimmy reached over and grabbed Bob’s moldy old half eaten electron sandwich
A Day in the Life
Once there was a atom named Trip and Daniel. There were both in search of an electron house.